Medicines Discovery Catapult and the BioIndustry Association stated in their 2018 State of the Discovery Nation Report, that:
“… over 80% of SMEs surveyed agreed that access to biosamples is hugely important for commercial development” and “… as many as 80% found accessing UK samples unexpectedly difficult…”
A major recommendation of this report was that actions need to be taken to improve sample and data access for all translational scientists. Hopefully, the information provided in this article will provide some assistance in this respect.
Understanding the Different Routes of Access
Most biosamples originate in the public sector in hospitals, clinics and collection centres. They are stored in biobanks, which may be called academic biobanks, hospital-based biobanks, or population biobanks.
For researchers in the private sector, there are two main ways of accessing human biosamples for research. Firstly, there is the direct route, which involves obtaining samples from biobanks. Secondly, there is the indirect route, which involves paying a commercial intermediary organisation to source the biosamples for you. These two routes are shown in this diagram:
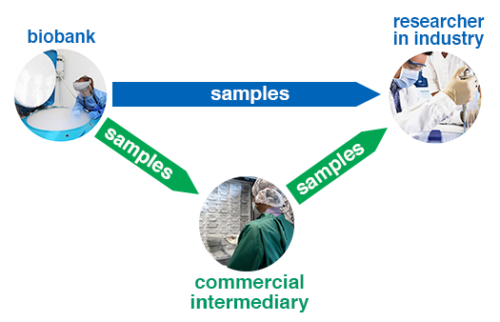
The Direct Route
To take the direct route, you need to identify a public sector biobank that is willing to supply the kind of samples you need.
A good place to start looking is the UKCRC Tissue Directory – an online directory, which allows you to search for biobanks with the samples you need, or biobanks that can start a collection for you. If your search is not successful, you may want to try the BBMRI-ERIC Directory, a similar but larger directory. These directories will provide the contact details you need to explore further and decide whether the biobank you have found is a good fit.
Whether you find a suitable biobank by this route will depend very much on what samples you are trying to source. If you are searching for a common disease type, with simple clinical information, you may find a biobank very easily. On the other hand, if you are searching for a rare disease, or you need extensive clinical information, your search may take much longer.
If you need samples to be collected in a very specific way, you may need to find a biobank that can start a prospective collection for you. In this case, there is an additional time delay while you wait for your collection to be completed.
Another important factor is whether the biobank’s access policy will allow industry access and, if they do, what are the conditions. In some cases, biobanks will only allow access to the private sector if they are involved in a research collaboration with the institution to which the biobank belongs. This information is frequently not advertised publicly, as reported recently by Langhof et al (2017). So, before investing any time in an application, it is best to contact the biobank to ensure that access will be possible on acceptable terms.
You also need to bear in mind that, there will be a cost recovery fee for samples. This covers the cost of all the technical and administrative work needed to collect, process, store and provide the biosamples and associated data.
Advantages of the Direct Route
- Better traceability – having chosen the source biobank, you are in direct contact with the team who collected the sample, and so if at any time you need to obtain additional information about your samples it will be easier to do so.
Disadvantages of the Direct Route
- Lengthy administrative processes – academic institutions often take a long time for administrative processes.
- Access policies – there is the question of whether the biobank’s access policies will permit industry access on acceptable terms. This needs to be checked early on.
The Indirect Route
Commercial intermediaries will generally be either:
- Commercial biobanks, which have established their own in-house stocks of biosamples by sourcing from public sector biobanks, and sometimes from other commercial intermediaries.
- Virtual biobanks or brokers, which do not keep any stock but liaise between biobank providers and the client requesting samples.
- CROs, which provide a variety of services often linked with clinical trials. Find out more about commercial intermediaries in my article The Main Actors Providing Biosamples To Industry.
Advantages of the indirect route
- Simpler administrative processes – commercial intermediaries may have simpler administrative processes and may not even require a material transfer agreement.
- More rapid supply – supply should be fast if a commercial biobank has the samples you need in stock. If not, then they can call on their well-established networks of biobank providers. They will know from experience which providers are prompt and reliable, so delays are less likely.
- Favourable access policies – commercial intermediaries are open to industry and do not require a collaborative research project.
Disadvantages of the indirect route
- Less traceability – commercial intermediaries may be reluctant to reveal the source of their samples because they want to protect their business relationship with both sample provider and client. The level of traceability allowed will vary from one commercial intermediary to another.
Cost considerations
The commercial intermediary needs to charge for the sourcing service it provides. Sometimes this means that commercial intermediaries are more expensive. However if, for example, samples have been sourced from overseas, they may come from countries where salaries and cost of living are lower. In such cases the cost recovery fee for sample processing will be reduced and hence the overall fee may also be reduced.
Final thoughts
Whether you should choose the direct route or the indirect route to source your biosamples will depend on a number of different factors including:
- How difficult it is to find the specific samples and associated information that you need.
- The amount of time you can allow for the search.
- Your budget and the relative costs of different sources.
- The importance of traceability for your work.
- Your corporate policy on procurement of biospecimens.
Hopefully, by weighing up the advantages and disadvantages described in this guide, you will find the access route that works best to meet your company’s biosample needs.
Dr Robert Hewitt is a Biobanking Specialist.
Previously, Robert worked in cancer research, both as a lecturer in pathology, at the University of Nottingham; and as a visiting fellow, at the National Institutes of Health. Since 1998, he has established/managed biobanks in London, Saudi Arabia, Singapore and Luxembourg. He was President of ISBER in 2007 and moved on to co-found ESBB in 2010, serving as the sole employee until 2017. After one year of industry experience, working for Trans-Hit Biomarkers, Robert now works independently as a consultant, promoting cooperation between academic biobanks and industry. He is active on LinkedIn and runs the ‘Drug Targets & Biomarkers’ group.







