MDC Connects is a series of informative webinars covering the drug discovery process, from our own experts and experts at our CRO network partners.
For a molecule to be worthy of entering preclinical development it needs to have the desired biological activity, as well as DMPK properties and a safety profile appropriate for the targeted therapeutic indication. In this phase of the drug discovery process biologists and chemists work to optimise the properties of the compounds by utilising computational modelling, chemical reactions and synthetic transformations and a suite of biological in vitro assays and in vivo models.
In this webinar we will focus on the methodologies used to establish and optimise the DMPK properties and biodistribution of the molecules. Firstly, Al Dossetter from MedChemica describes how multiple data sources can be used to improve the properties of compounds using in silico drug design and how this method can lead to a good quality candidate drug in fewer iterations. Richard Weaver from XenoGesis outlines examples of DMPK properties of the molecule, how they are assessed, and common mistakes made, providing illustrations of how the issues can be resolved. Finally, Juliana Maynard from MDC describes how imaging technologies can be used to establish the biodistribution and accumulation of the compounds in vivo.
Recording

Slides
In-Silico Drug Design – what to do, what not to do – project driven examples at MedChemica
Optimising ADME and PK properties: Common mistakes made and how to identify and resolve the key issues at XenoGesis
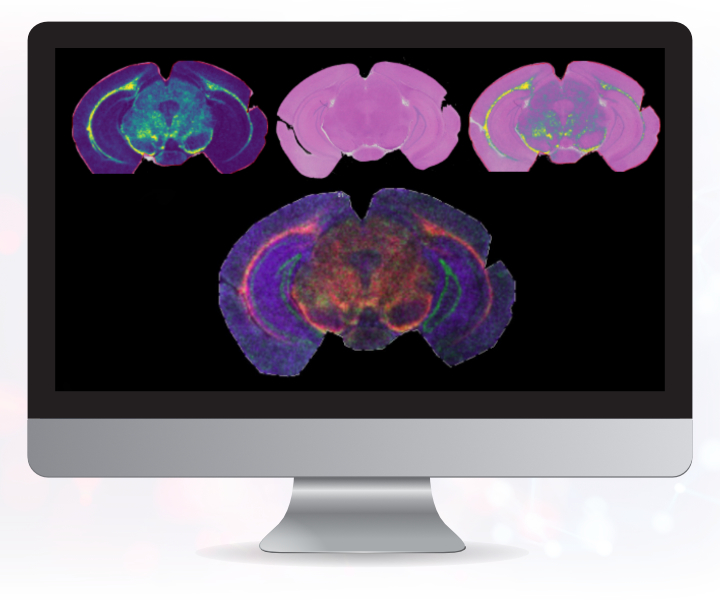
Imaging technologies to understand the pharmacokinetics and biodistribution of a candidate compound at MDC