Health Data Research UK (HDRUK) is the national institute for health data science whose vision is to unite the UK’s health data to enable discoveries that improve peoples’ lives. Launched in April 2018, HDR UK works in partnership with the NHS, industry, academia and patients, and aims to help better understand diseases and discover new ways to prevent, treat and cure them through safe and secure access to health data.
Integral to this endeavor is the formation of seven Health Data Research Hubs aligned to disease focused datasets, clinical trials and real-world evidence. Through the curation and linkage of different types of health data, whilst critically maintaining strict controls around data privacy and consent, the Hubs will help to make health data more accessible to researchers and innovators. This aims to speed up research for new medicines and technologies that support quicker diagnoses, management and improve patient outcomes.
Introducing the UK’s Health Data Research Hubs:
| BREATHE | Health Data Research Hub for Respiratory Health |
| DATA-CAN | Health Data Research Hub for Cancer |
| Discover-NOW | Health Data Research Hub for Real World Evidence |
| Gut Reaction | Health Data Research Hub for Inflammatory Bowel Disease |
| INSIGHT | Health Data Research Hub for Eye Health |
| NHS DigiTrial | Health Data Research Hub for Clinical Trials |
| PIONEER | Health Data Research Hub for Acute Care |
Industry engagement
Funded by the Industrial Strategy Challenge Fund, the Hub programme was developed in response to the Life Sciences Industrial Strategy and successful engagement with industry is fundamental to its success.
As part of Medicines Discovery Catapult’s support of the drug discovery SME community a recent workshop was held to showcase two of the hubs: PIONEER – the Health Data Research Hub for acute care, and INSIGHT – the Health Data Research Hub for eye health.
PIONEER
Acute care episodes are rising and place a huge burden on the NHS. It is an area of great unmet need with rising costs, diagnostic delay, poor health outcomes and over-stretched front door services.
PIONEER will be the single, largest acute care medical database globally, comprising of electronic health records for more than one million acute episodes including over 24 million prescriptions, 268 million investigations and 25 million procedures, captured by one of the most advanced hospital digital systems in the world developed by an award-winning team at University Hospitals Birmingham NHS Foundation Trust.
PIONEER aims to individually link data across acute health care providers including primary care, out of hours, ambulance, secondary care and community care to be able to track health care journeys from onset to outcome. This integrated view of acute care challenges will provide a platform for collaborative working across clinical, data science, Information Governance, academia and industry to help meet HDR UK, ISCF and NHS priorities. The vision is that this system of working, established with regional depth could eventually be scaled for national impact.
Examples of end uses for research using acute care data:
- New devices
- Healthcare AI
- New ways to deliver healthcare
- Patient stratification
- Earlier diagnosis
- Healthy ageing
- Helping patients manage their own health
INSIGHT
NHS Digital statistics in 2018 revealed that ophthalmology now surpasses orthopaedics in annual outpatient attendance figures.
The INSIGHT Health Data Research Hub is focused on eye disease but also its application to wider health, including diabetes and dementia. It will use anonymised large-scale data and advanced analytics, including artificial intelligence, to develop new insights in disease detection, diagnosis, treatments and personalised healthcare.
The Hub will unlock opportunities for research using large patient datasets from Moorfields Eye Hospital and University Hospitals Birmingham NHS Foundation Trust, with 8 million retinal photographs and 3 million Optical Coherence Tomography images, many offering over 10 years of longitudinal tracking. The Hub will provide a platform to learn from the real world, not only for blinding eye health but also for systemic disease particularly in neurological and cardiovascular conditions.
Workshop Outputs
The workshop, held on 6th February at The Alderley Park Conference Centre, brought together representatives from across industry, MDC and HDR UK, with PIONEER and INSIGHT Hub leaders and experts. As well as hearing in detail about the the vision and aims of the two Hubs, attendees also heard more about the HDR UK infrastructure: the UK Health Data Research Alliance, the Health Data Research Hub programme, and newly launched Health Data Research Innovation Gateway Minimum Viable Product (MVP)
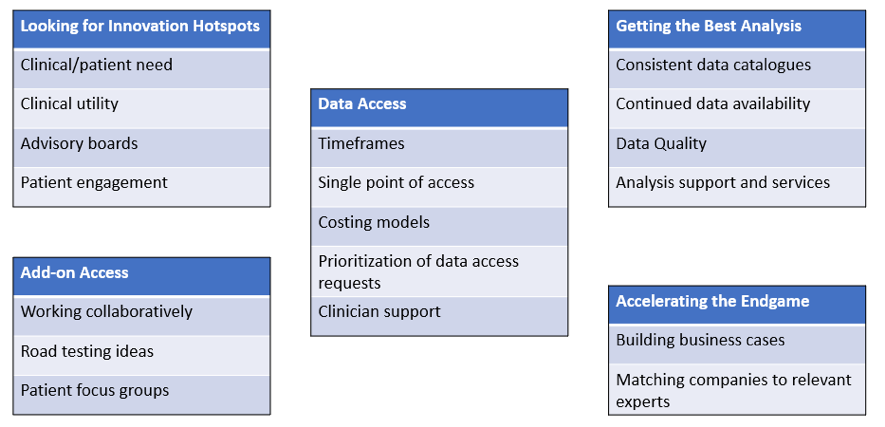
An interactive session offered the opportunity for all attendees to discuss five key themes for getting the best out of industry engagement with the PIONEER and INSIGHT Hubs (Fig 1).
Fig 1. Workshop discussion topics
There was a tremendous level of engagement and energy from everyone in the room and lots of extremely useful discussion! In addition to the key points raised on the tables illustrated above, we covered topics such as prioritisation of emerging challenges; learning from other UK investments in this space; future opportunities such as integration of wider datasets; and sharing of information, outputs and systems with those who need it most, including frontline services and GPs.
As the Health Data Research Hubs develop their strategy over the coming months, successful industry engagement will help strike a balance for innovation to bring commercial benefit, whilst making a difference to the NHS and enhancing patients’ lives.
If you want to join the discussion, please do get in touch:
- Health Data Research UK: enquiries@hdruk.ac.uk
- PIONEER Health Data Research Hub: pioneer@contacts.bham.ac.uk
- INSIGHT Health Data Research Hub: enquiries@insight.hdrhub.org
About the author
Joanne Hartley is a Samples & Data Access Executive with over 20 years’ experience in life sciences, having held several research roles at AstraZeneca both in the lab and as a computational biologist. She leads Medicines Discovery Catapult’s work to help drug discovery companies access the clinical data they need to develop novel medicines faster.